When encountering exogenous antigens, T cells that express receptors capable of binding to a specifically compatible peptide–MHC (pMHC) complex will expand, resulting in a massive population of antigen-specific T cells that initiate the adaptive immune response ( 7– 11). The number of each lymphocyte clone changes dramatically and depends on cell specificity and the history of antigen exposure. The formation and revision of the T and B cell lymphocyte receptor repertoire is a highly dynamic process. Construction of the TCR with an alpha chain and a beta chain is also a process that contributes to receptor diversity. 
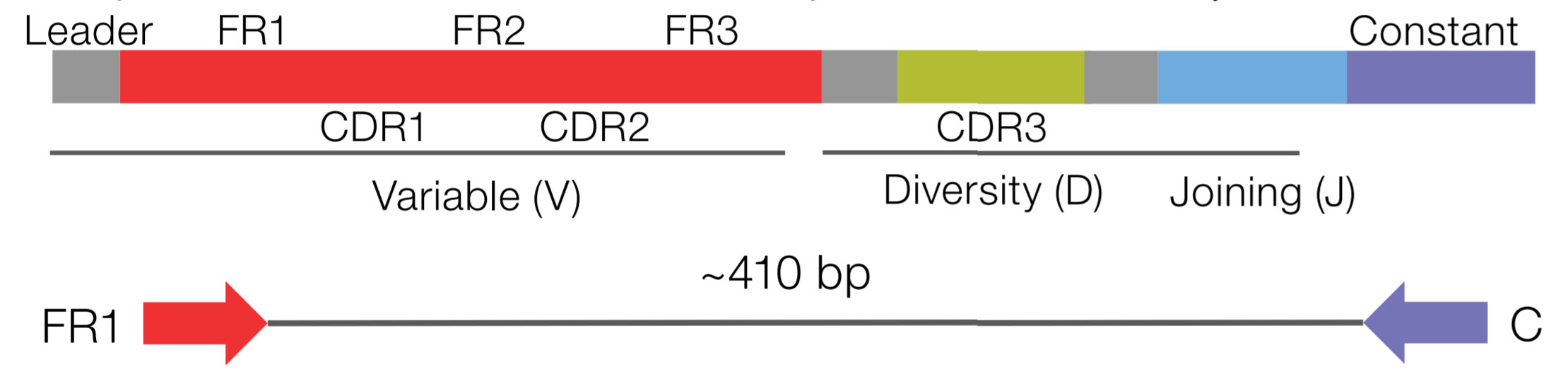
Thus, CDR3 is the most diverse component of a receptor, which binds MHC molecules and (or) antigens. While CDR1 and CDR2 are formed by variable (V) gene, CDR3 is generated by random selection and recombination of variable (V), diversity (D), and joining (J) gene segments in the heavy chain (V and J region gene segments in light chain) ( 5, 6) (Figure 1). CDRs are the variable portion of the receptor and determine the antigen specificity. For T cells, the variable region of each TCR chain consists of three complementary determining regions (CDRs) and four frame regions (FRs). This repertoire is generated by a complex series of genetic events ( 4). Generation of a Diverse Immune RepertoireĪmazing diversity makes the immune system the most effective system to fight against a broad scope of disease causing pathogens. We then focus on the success of this technology in facilitating the exploration of infection-related immune repertoires for clinical diagnosis, treatment, and prevention. In this review, we introduce the implementation of HTS to the study of the immune repertoire and review the associated bioinformatic tools required for data processing and analysis. These methods create an unprecedentedly high-resolution picture of the immune repertoire and also provide massive data that cover each lymphocyte from the sample, in theory, dispensing with limitation of sequencing quantity ( 3).Ĭonsidering the extremely important role of the adaptive immune system in defending against infectious agents, HTS has great potential to aid in the discovery novel infectious agents and also offers new approaches for antibody or vaccine development. During the past two decades, however, technical advances in high-throughput sequencing (HTS), also known as next-generation sequencing (NGS), along with evolving bioinformatic and statistical tools, have provided a new approach capable of analyzing the immune repertoire at the single sequence level. However, these low-throughput techniques lack the power to provide a broad picture of the full immune repertoire. Several sequencing strategies, for example, Sanger sequencing, have been implemented to determine cDNA segments encoding variable regions of immunoglobulin (or TCRs) ( 1, 2). Thus, study of the immune repertoire, portrayed as the antigen-specific information within lymphocytes, has been a key to understanding the response of adaptive immunity during infection.ĭespite extensive efforts using traditional techniques, analysis of the immune repertoire with high resolution has remained difficult. This profound diversity of T (TCRs) and B cell receptors (BCRs) is generated by V–D–J gene recombination of the TCR/BCR locus and subsequent somatic hypermutation and class-switching recombination of B cells after antigen stimulation. The foundation of the adaptive immune response is based on the enormous diversity of T and B cell antigen receptors that can recognize epitopes from a near infinite number of different internal and external antigens. The adaptive immune system is composed of B and T cells that form a highly selective guard against evolving pathogens. This progress in methodology enhances the understanding of immunologic changes during pathogen challenge and also provides a basis for further development of novel diagnostic markers, immunotherapies, and vaccines. HTS techniques enable the determination of complementary regions of lymphocyte receptors with unprecedented efficiency and scale. In this article, we review the recent advances in immune repertoire study of infectious diseases, which were achieved by traditional techniques and high-throughput sequencing (HTS) techniques. The immune repertoire, the collection of T and B cells with functional diversity in the circulatory system at any given time, is dynamic and reflects the essence of immune selectivity. The selectivity of the adaptive immune response is based on the enormous diversity of T and B cell antigen-specific receptors. 2Department of Medicine, Division of Pulmonary and Critical Care Medicine, University of California San Francisco, San Francisco, CA, USA.1Department of Pulmonary Medicine, Zhongshan Hospital, Fudan University, Shanghai, China.Dongni Hou 1 Cuicui Chen 1 Eric John Seely 2 Shujing Chen 1 Yuanlin Song 1* 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
